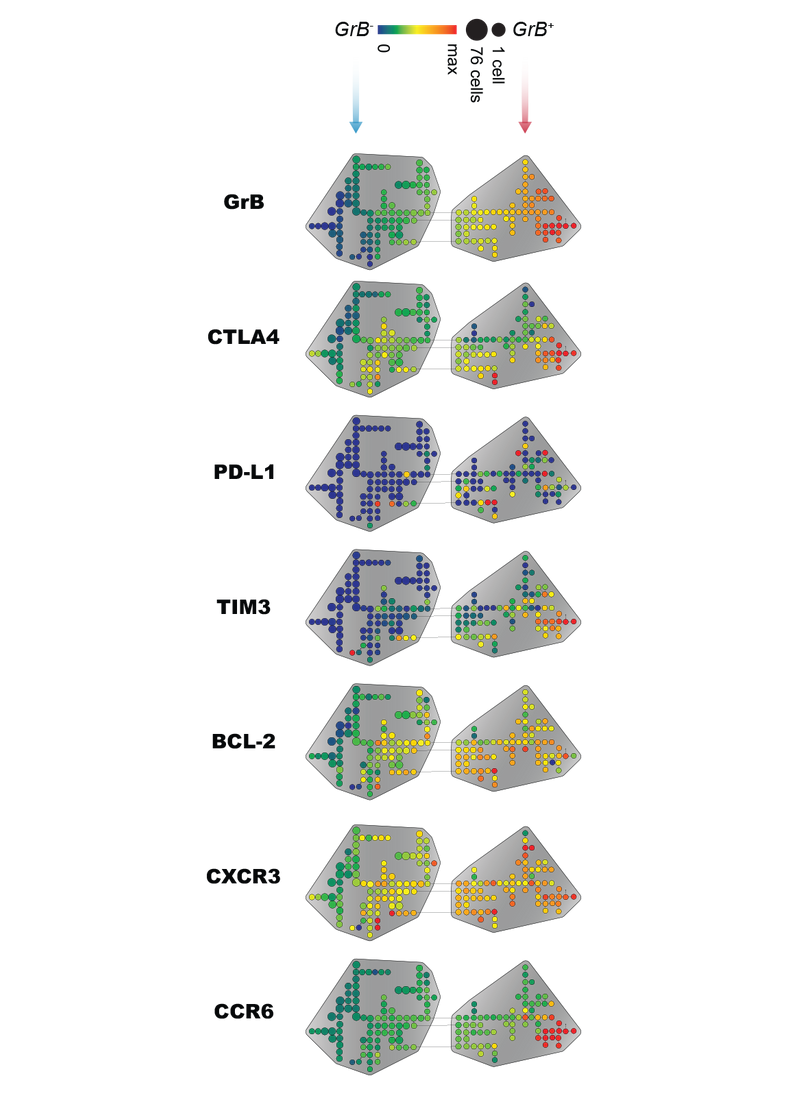
Our immune system acts in a very specific and elegant way against foreign antigens such as infections, solid organ transplants and even cancer. For example, each clone of T cells out of a billion we own, recognizes one foreign antigen and reacts only against it, while the rest of the billion T cell clones remain inactive. Moreover, our immune system balances two opposing forces, the effector arm that attacks antigens and the regulatory arm that is protective.
Current immunosuppressive drugs (ISD) inhibit the whole immune system including both the effector and the protective regulatory axes of adaptive immunity, putting patients at high risk for complications longterm. Failure to promote the regulatory arm of the immune system leads to chronic rejection and worse long-term outcome of solid organ transplant recipients.
A major focus in our lab currently is regulatory T cells and their activation induced cell death. The goal is to use this knowledge to develop therapies that promote the regulatory arm of the immune system to protect against autoimmunity and rejection in solid organ transplant. We are also performing drug screening to discover drugs that preferentially promote Tregs while suppressing the effector arm of the immune system. Another focus is to engineer Regulatory T cells to improve their therapeutic potentials as Cell therapy.
Another focus of our laboratory is to engineer regulatory T cells using T cell responsive cytokine delivery platform and CAR-Tregs. Our work has been highlighted in the “What’s Hot What’s New” sessions at the American Transplant Congress over the last few years. We also recently discovered a key role for Qa1 restricted regulatory T cells in controlling antibody-mediated rejection in transplantation.
We recently described in collaboration with Ghobriel Lab at Dana Farber Cancer Institute that one of the major immunosuppressive mechanisms promoting myeloma progression is the expansion of regulatory T cells (Tregs) in the bone marrow microenvironment. Further analysis of the interaction between myeloma cells and Tregs using gene sequencing and enrichment analysis uncovered a feedback loop, wherein myeloma-cell-secreted type 1 IFN induced proliferation and expansion of Tregs. Using bone marrow (BM) aspirates of MM patients, we found that Tregs were expanded and activated in the BM microenvironment at early stages of myeloma development and that Type 1 IFN signaling is associated with Treg activation and overall patient survival. Our results thus suggest that blocking type 1 IFN signaling represents a potential strategy to target immunosuppressive Treg function in MM.
Another project with Ghobriel Lab explored the role of macrophages in anti-myeloma activity. This work highlighted the importance of the immune system in controlling tumor progression.
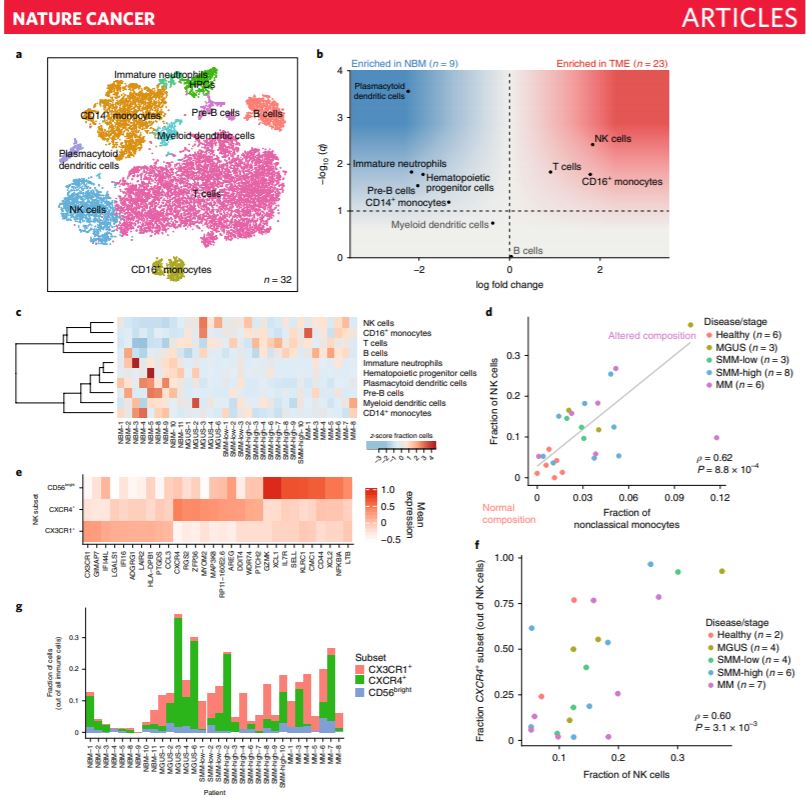
Using single-cell RNA sequencing of patient samples, we revealed the role of the compromised immune microenvironment in the precursor stages of multiple myeloma (zavidij et al. Nature Cancer).
We are currently applying those approaches to other rare tumors such as Chordoma
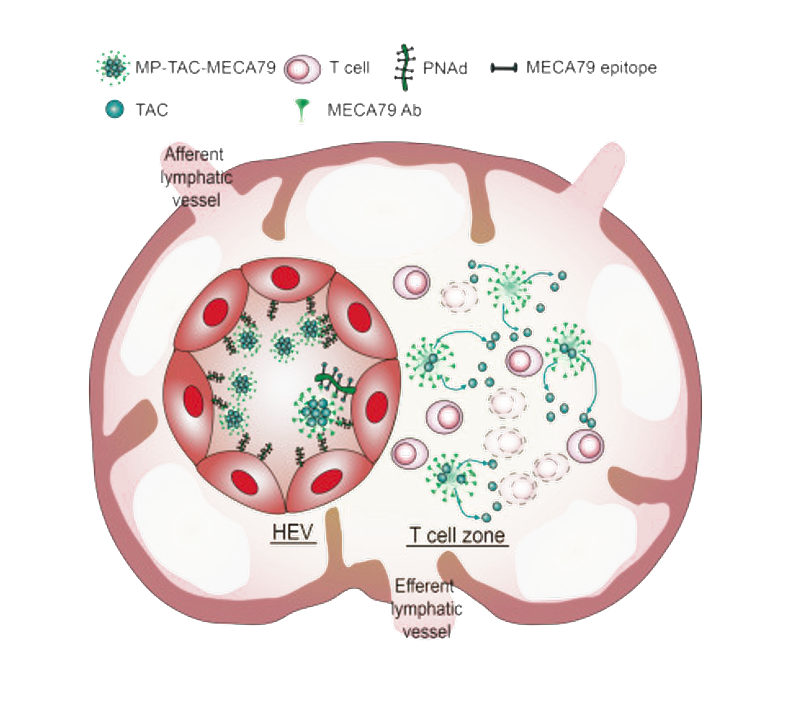
Our lab aims to harness the advancements in nanotechnology to develop novel therapies to different immune mediated diseases and to revolutionize the field focusing on antigen specific therapies and drug delivery to the areas of inflammation in auto and allo-immunity. We are conducting this work in collaboration with pioneering laboratories at MIT and Harvard. Nanotechnology is emerging today at the forefront of science and medicine. Nanotechnology brings a focus on development of more effective therapeutic systems. These “smart therapeutics” have the potential of targeting specific organs and or antigen specific auto-reactive or allo-reactive cells while avoiding the toxicity of conventional immunosuppressive medications. This field of targeted drug delivery through nanotechnology is in its infancy and our work in transplantation is pioneering this proof of concept. We are also engineering cell therapy to apply it to different immunologic mediated diseases.
In collaboration with Gang Lin and Carl Nathan at Cornell, we showed that the ubiquitously expressed constitutional proteasome is replaced by the immunoproteasome mainly in immune cells during activation by allo-antigens. Targeting the immunopreatosome provides a unique opportunity to develop more targeted therapy of the immune system. Indeed, this multidisciplinary team in collaboration with Dr. Huilin Li reported the crystal structure of the immunoproteasome and exploited the differences with the constitutional proteasome to design and synthesize small specific inhibitors of the immunoproteasome. These new inhibitors promise to revolutionize the treatment of antibody-mediated rejection (AMR) in solid organ transplant, a critical unmet need in our field.
In the absence of non-invasive tests to accurately predict kidney transplant rejection and to monitor the immune system, we are performing translational research to develop genomic and proteomic biomarkers. Those biomarkers will help us tailor the immunosuppressive medications to patient’s need and help reduce complications. Our work pioneered the development of urinary Extracellular Vesicles as a non-invasive diagnostic means to monitor and evaluate kidney transplant recipients.
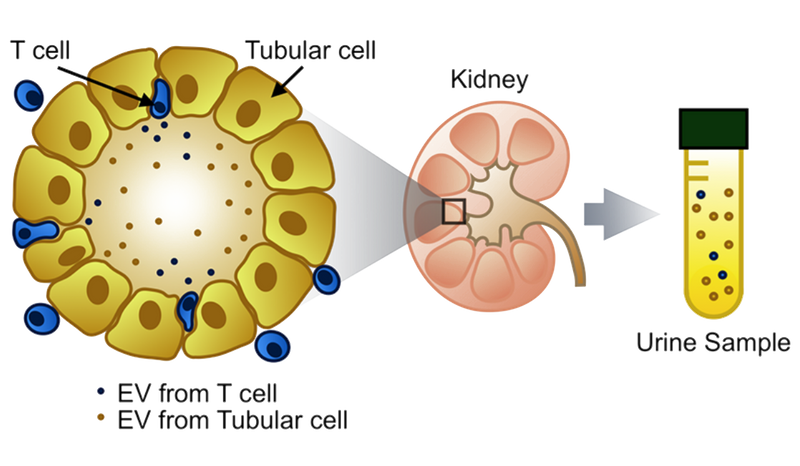

In collaboration with other investigators in Boston, we created a biobank that collects samples of bodily fluid and tissue from kidney transplant recipients with the goal of improving our understanding of the process of rejection. Those samples will be preserved so that long term future research can be carried out.
We have established uniform protocols for all stages of processing of cells for use in functional assays (ELISPOT, Multiplex Luminex, Flow Cytometry, Mass Cytometry, etc). This includes establishing protocols for sample collection (types of tubes), handling, freezing, thawing, shipping, and conditions for storing (length of time, freezing). Thus, for all stages of cell isolation, preparation, freezing, storing and thawing we have adopted standard procedures. We have developed standard operating procedures for isolation, storage and analysis of RNA from graft biopsies and patient peripheral blood and urine. Similarly, we developed techniques to isolate microvesicles from peripheral blood and urine. This platform is promising for developing new biomarkers for immunologic diseases.
We have also began growing a liquid biopsy consortium in collaboration with those who share our vision of advancing precision medicine for patients with kidney diseases and kidney transplant recipients through non-invasive techniques. Find out more at the website below!
The Azzi lab focuses on promoting comprehensive training for the next generation of medical professionals across multiple disciplines related to immune mediated diseases. Our innovative approach to training aims to foster the collaborative spirit of our future scientists who promises to transform the world we live in.
Our vision is to bring people from different ethnicities, race, gender and religious believes from all over the globe to work together in developing a better world through innovative discoveries that serve all humanity.